- Basic Information
- Market and Price
- Images and Vedios
1. Product Summary of Potassium Fluoride

Product Name: Potassium Fluoride
Synonyms: Anhydrous Potassium Fluoride
CAS NO.: 7789-23-3
Molecular Weight: 58.10
Molecular Formula: KF
CBNumber: CB4237549
Saturated vapor pressure (kPa):133.3Pa (885 DEG C)
Solubility: soluble in water, hydrofluoric acid, liquid ammonia, insoluble in alcohol.
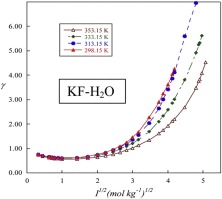
Solubility in water: 44.7g/0 DEG C; 53.5g/10 DEG C; 94.9g/20 DEG C; 108g/30 DEG C; 138g/40 DEG C; 142g/60 DEG C; 150g/80 DEG C;
Single isotope mass: 57.96211 Da
Nominal mass: 58 Da
Average quality: 58.0967 Da
Usage: In the metal finishing, batteries, coatings, photographic chemicals, industrial chemicals. Package: Our potassium fluoride is packaged with aluminum foil bag (20kg/bag) or cardboard drum (75kg/drum), or will be packed upon clients’ requests.
Package: 1)20 kg /aluminum foil bag or 75kg/drum (for spray dried KF) 2)25 kg /aluminum foil bag or 25 kg/drum (for Granular KF) 3)1000 L/drum or 20 MT/ISO Tank (for KF,40% solution) Xinxiang Yellow River Fine Chemical Industry Co., Ltd. is the largest manufacturer of Potassium Fluoride in the world. We produce the full series of Potassium Fluoride including spray dried KF, granular KF and KF solution.
2. Basic Information of Potassium Fluoride
Potassium Fluoride, with its CAS registry number 7789-23-3, has other name of Fluorure de potassium. With its molecular formula of KF and molecular weight of 58.1, it has its EINECS number of 232-151-5. Being a kind of crystalline powder with melting point of 858℃, boiling point of 1505℃, KF is soluble in hydrofluoric acid and liquid ammonia while slightly soluble in alcohol and acetone.
Potassium Fluoride has two hydrate compounds, KF•2H2O and KF•4H2O. With a good solubility in water, it absorbs moisture easily, so this chemical shall be stored in sealed container. When comes to its storage, it should be kept in a tightly closed container in a cool, dry, ventilated area, away from acids and alkalis. Besides, this chemical is incompatible with platinum plus bromine trifluoride, and it could react with strong acids to form hydrogen fluoride.
In addition, Potassium Fluoride is widely applied in various fields. KF is a commen raw material for inorganic fluorine industry and can widely be used in the production of agrochemical and pharmaceutical intermediates. KF is also used as metal surface treatment products in electronic industry. In fluorine deficiency area, KF is added to the table salt as a kind of food additive. Moreover, KF can be used in the production of flux products, and also used as preservative, catalyzer and water absorbing agent.
1). Properties of Potassium Fluoride
The other characteristics of Potassium Fluoride (CAS NO.: 7789-23-3) can be summarized as: (1). Specific Gravity: 0.3-0.6; (2) Boiling Point: 1505℃ (2741F); (4) Melting Point: 860℃ (1580F); (5) Vapor Density (Air=1): 2.0.
2). Preparation and Use
Preparation: Potassium Fluoride could be produced through the neutralization method, with the following chemical equation:KOH+HF→KF+H2O.
Use: Potassium Fluoride is widely applied in the preparation for various kinds of organic fluoride. For example, KF could used to promote the processing of the following carbonyl-coupling reaction:
3). Hazard identification of Potassium Fluoride and its according first aid measures:
Hazard identification of Potassium Fluoride are as below:
Skin Contact: Causes severe irritation and possibly burns to the skin. May be absorbed through the skin. Effects may not appear immediately.
Eye Contact: Causes irritation. May be extremely irritating with possible burns to eye tissue and permanent eye damage may result.
Inhalation: May cause irritation and burns to the respiratory tract. May be absorbed through inhalation of dust. Irritation and burning effects may not appear immediately.
Ingestion: May cause salivation, nausea, vomiting, diarrhea, and abdominal pain, followed by weakness, tremors, shallow respiration, cardopedal spasm, convulsions, and coma. May cause brain and kidney damage. Death may be caused by respiratory paralysis. Affects heart and circulatory system.
Chronic Exposure: Chronic exposure may cause mottling of teeth and bone damage (osteosclerosis) and fluorosis. Symptoms of fluorisis include brittle bones,weight loss, anemia, calcified ligaments, general ill health and joint stiffness.
Aggravation of Pre-existing Conditions: Populations that appear to be at increased risk from the effects of fluoride are individuals that suffer from diabetes insipidus or some forms of renal impairment.
First aid procedures for contacting Potassium Fluoride should be pre-planned for fluoride compound emergencies.
Skin Contact: Promptly wash with soap and water, rinsing until clean. Remove contaminated clothing and wash before reuse. CALL A PHYSICIAN IMMEDIATELY.
Eye Contact: Immediately flush eyes with plenty of water for at least 15 minutes, lifting lower and upper eyelids occasionally. Get medical attention immediately.
Inhalation: If inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. CALL A PHYSICIAN IMMEDIATELY.
Ingestion: Administer milk, chewable calcium carbonate tablets or milk of magnesia. Never give anything by mouth to an unconscious person. CALL A PHYSICIAN IMMEDIATELY.
Note to Physician: For large exposures, systemic effects (hypocalcemia and hypomagnesia) may occur. Potassium can reduce blood pressure and cause coma.
4). Exposure Controls/Personal Protection
The exposure controls and personal protection of Potassium Fluoride are as below:
Airborne Exposure Limits: - OSHA Permissible Exposure Limit (PEL): 2.5 mg (F)/m3 (TWA) - ACGIH Threshold Limit Value (TLV): 2.5 mg (F)/m3 (TWA)
Ventilation System: A system of local and/or general exhaust is recommended to keep employee exposures below the Airborne Exposure Limits. Local exhaust ventilation is generally preferred because it can control the emissions of the contaminant at its source, preventing dispersion of it into the general work area. Please refer to the ACGIH document, Industrial Ventilation, A Manual of Recommended Practices, most recent edition, for details.
Personal Respirators (NIOSH Approved): If the exposure limit is exceeded, a half-face dust/mist respirator may be worn for up to ten times the exposure limit or the maximum use concentration specified by the appropriate regulatory agency or respirator supplier, whichever is lowest. A full-face piece dust/mist respirator may be worn up to 50 times the exposure limit, or the maximum use concentration specified by the appropriate regulatory agency, or respirator supplier, whichever is lowest. For emergencies or instances where the exposure levels are not known, use a full-facepiece positive-pressure, air-supplied respirator. WARNING: Air-purifying respirators do not protect workers in oxygen-deficient atmospheres.
Skin Protection: If dealing with Potassium Fluoride, wear impervious protective clothing, including boots, gloves, lab coat, apron or coveralls, as appropriate, to prevent skin contact.
Eye Protection: If contact with Potassium Fluoride, use chemical safety goggles and/or full face shield where dusting or splashing of solutions is possible. Maintain eye wash fountain and quick-drench facilities in work area.
In addition, if in the event of a fire, wear full protective clothing and NIOSH-approved self-contained breathing apparatus with full facepiece operated in the pressure demand or other positive pressure mode.
5). Descriptors of Structure of Potassium Fluoride
(1)SMILES:[F-].[K+]
(2)Std. InChI:InChI=1S/FH.K/h1H;/q;+1/p-1
(3)Std. InChIKey:NROKBHXJSPEDAR-UHFFFAOYSA-M
6). Toxicity Information of Potassium Fluoride
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| frog | LDLo | subcutaneous | 420mg/kg (420mg/kg) | Comptes Rendus des Seances de la Societe de Biologie et de Ses Filiales. Vol. 124, Pg. 133, 1937. | |
| guinea pig | LDLo | oral | 250mg/kg (250mg/kg) | "Merck Index; an Encyclopedia of Chemicals, Drugs, and Biologicals", 11th ed., Rahway, NJ 07065, Merck & Co., Inc. 1989Vol. 11, Pg. 1214, 1989. | |
| mouse | LD50 | intraperitoneal | 40030ug/kg (40.03mg/kg) | Deutsche Zahnaerztliche Zeitschrift. Vol. 34, Pg. 484, 1979. | |
| rat | LD50 | intraperitoneal | 64mg/kg (64mg/kg) | U.S. Atomic Energy Commission, University of Rochester, Research and Development Reports. Vol. UR-154, Pg. 1951, | |
| frog | LD50 | oral | 245mg/kg (245mg/kg) | U.S. Atomic Energy Commission, University of Rochester, Research and Development Reports. Vol. UR-154, Pg. 1951, |
potassium fluoride solution Product Overview
Solvay Fluorides, LLC does not sell potassium fluoride solution (KF) directly to consumers.Most potassium fluoride solution is used in industrial applications and processes.
3. Product Uses
Potassium fluoride solution is used for pH adjustment in industrial textile processing facilities or laundries. It is also an intermediate or raw material used in other chemical synthesis or processes;most often for agrichemical or pesticide products. Recently, potassium fluoride has been used to promote the synthesis of 1,2-oxazoline derivative, fluorination, Suzuki coupling and synthesis of thiophene derivatives.
- For glass engraving, food preservation, plating.
- Can be used as welding flux, insecticide, catalyst, absorbent (absorb HF and moisture).
- Can be used as the fluorinated agent of organic compounds.
Most often for agrichemical or pesticide products. Potassium fluoride is sold in an aqueous (water)solution.Potassium fluoride solution is corrosive and contact can severely irritate and burn the skin or eyes causing possible eye damage. Breathing potassium fluoride solution vapors can irritate and burn the nose, throat, and lungs. Chronic exposure to KF may cause nosebleeds, cough, wheezing and shortness of breath. When heated potassium fluoride solution releases hydrogen fluoride, a toxic,corrosive gas.
Inhalation or ingestion of large amounts of potassium fluoride solution can cause nausea, vomiting and loss of appetite. Exposure to high concentrations or long term exposure can cause fluoride poisoning with stomach pain, weakness, convulsions and death. Long term or repeated exposures can cause deposits of fluorides in bones and teeth, a condition called fluorosis. Fluorosis may cause pain, disability and discoloration of teeth.
Potassium fluoride Uses References
《Potassium fluoride improving the ignition and combustion performance of micron-sized aluminum particles in high temperature water vapor》, Wei Shi,Baoxin Dai,Baozhong Zhu,Yunlan Sun &Ying Chen, Energy Sources, Part A: Recovery, Utilization, and Environmental Effects Volume 45, 2023 - Issue 2
To improve the ignition and combustion characteristics and combustion efficiency of micron-sized aluminum particle in high temperature water vapor atmosphere, a self-designed tube furnace setup was used to study the effects of potassium fluoride (KF) on the ignition and combustion characteristics of micron-sized aluminum particle in high temperature water vapor.
《Effect of silver diamine fluoride/potassium iodide treatment on the prevention of dental erosion in primary teeth: an in vitro study, Lamia Khairy Gadallah》, Engie Mohamed Safwat, Reham Sayed Saleh, Shereen Musa Azab & Maha Moussa Azab , BDJ Open volume 9, Article number: 24 (2023)
The application of potassium iodide KI following SDF was supposed to reduce the potential staining caused by SDF application by scavenging the free silver ions and forming a creamy white silver iodide precipitate.
Exposure Potential
Workplace Exposure - Potassium fluoride is corrosive and toxic by ingestion, inhalation or contact with skin and eyes. Exposures can occur at a potassium fluoride manufacturing facility or a manufacturing, packaging or storage facility that handles KF. Exposure may also occur in the event of a transportation incident. Persons involved in maintenance, sampling and testing activities, or in the loading and unloading of KF containers are at greater risk of exposure.
Following good industrial hygiene practices will minimize the likelihood of KF exposure;however, persons involved in higher risk activities should always wear proper personal
protective equipment such as rubber gloves and boots, goggles and a hard hat. In instances where the potential for splashes is high, a face shield should also be worn.
Exposure limits for potassium bifluoride (per OSHA, ACGIH, and other agencies) are listed as the “fluoride” content rather than as KBF specifically. Please consult the Safety Data Sheet for information concerning exposure limits.
Consumer Exposure to Products Containing Potassium Fluoride Solution - Solvay Fluorides, LLC does not sell potassium fluoride solution directly to consumers although it may be used in some consumer cleaning products. The user should use these products in strict adherence with the manufacturer’s use and/or label instructions.
Environmental Releases - Spills of potassium fluoride solution should be contained and isolated from waterways and sewers or drains. The contaminated area should be washed down with plenty of water. Lime or calcium hydroxide may be used to neutralize contaminated water and immobilize the fluoride ions as calcium fluoride. Disposal should be in accordance with applicable local, state or federal regulations. Persons attempting to clean up potassium fluoride solution spills should wear proper personal protective equipment (see guidelines in Workplace Exposure section of this document or Safety Data Sheet). If required, report spills to the appropriate state or federal authorities.Fires - Fires involving potassium fluoride solution should be extinguished using measures appropriate to the circumstances and surrounding environment. Hazardous decomposition products such as hydrogen fluoride vapor can be generated if KF is involved in a fire. Fire fighters should wear self-contained breathing apparatus and protective suits.
For additional information concerning potassium fluoride solution emergency response procedures,please consult the Safety Data Sheet.
Health Information
Concentrations of potassium fluoride solution typically found in consumer products may pose risk of symptoms due to skin, ingestion or inhalation exposure. Persons suffering from eye or ingestion exposure to consumer strength potassium fluoride solution products may experience symptoms similar to persons exposed to industrial strength potassium fluoride solution (see below).
Exposures to potassium fluoride solutions can produce the following adverse health affects:
Contact - Skin exposures can cause symptoms ranging from minor skin irritation to painful redness and swelling. Severe burns can occur if treatment is delayed after exposure to potassium fluoride solution. Eye exposure to potassium fluoride solution may result in severe eye irritation, burns or even blindness.
Inhalation - The inhalation of potassium fluoride solution vapors can cause symptoms ranging from nose and throat irritation to coughing and difficulty breathing. Aspiration may cause pulmonary edema (fluid on the lungs) and pneumonitis (inflammation of the lungs). Repeated or prolonged exposures may cause sore throat, nosebleeds and chronic bronchitis. Prolonged exposure may cause hypocalcemia (reduced calcium levels) with nervous problems (tetany)and cardiac arrhythmia (irregular heart beat) and /or spasms.
Ingestion - The ingestion of potassium fluoride solution may cause burns of the mouth and throat and perforation of the esophagus and stomach. Nausea, bloody vomiting, abdominal pain, diarrhea, difficulty breathing, swelling of the throat, loss of consciousness, coma and heartfailure can also occur.
Other Effects - The International Agency for Research on Cancer (IARC) has not determined sodium fluoride solution to be carcinogenic (cancer causing).For more information on health effects and routes of exposure, or for information concerning proper first aid measures, please consult the Safety Data Sheet.
Environmental Information
Potassium fluoride solution is not known to bioaccumulate or persist in the environment for more than a few days. For more ecological and environmental information concerning this product, please consult the Safety Data Sheet.
Physical Hazard Information
Potassium fluoride solution is corrosive and can corrode some metals. It is not flammable or explosive.Exposure of potassium fluoride solution to strong acids, strong bases, metals, glass or high temperatures can cause decomposition. Decomposition of potassium fluoride solution will result in the liberation of hydrogen fluoride gas.
For more information concerning the physical hazards of this product, please consult the Safety Data Sheet.
Regulatory Information
Regulations may exist that govern the manufacture, sale, export, import, storage, transportation,use and/or disposal of this chemical. These regulations can vary by city, state, country or geographic region. Information may be found by consulting the relevant Safety Data Sheet specific to your country or region.
Xinxiang Yellow River Fine Chemical Industry Co., Ltd. is the largest manufacturer of Potassium Fluoride in the world. Xinxiang Yellow River produce the full series of Potassium Fluoride including spray dried KF, granular KF and KF solution.Potassium fluoride is the chemical compound with formula KF [CAS: 7789-23-3]. KF is a crystalline powder with m.w. of 58.10, melting piont of 858℃, boiling point of 1505℃ and density of 2.481 g/cm3. KF is insolube in alcohol but have a good solubility in water. It absorbs moisture easily and shall be stored in sealed container. KF is the primary source of the fluoride ion for applications in chemical reactions. Aqueous solution of KF is alkaline and will etch glass. As a result, KF is a commen raw material for inorganic fluorine industry and can widely be used in the production of agrochemical and pharmaceutical intermediates. KF is also used as metal surface treatment products in electronic industry. In fluorine deficiency area, KF is added to the table salt as a kind of food additive. In addition, KF can be used in the production of flux products, and also used as preservative, catalyzer and water absorbing agent.
Our potassium fluoride is packaged with aluminum foil bag (20kg/bag) or cardboard drum (75kg/drum), or will be packed upon clients’ requests.